SOL-804
SOL-804 :
Treatment for metastatic prostate cancer
SOL-804 is a metastatic prostate cancer treatment being developed by Dyna Therapeutics, a subsidiary specializing in formulation development for Bukwang Pharmaceutical.Dyna Therapeutics, our Seoul-based subsidiary, is currently advancing SOL-804 as a promising candidate for the treatment of prostate cancer. We have secured global development and commercialization rights for SOL-804 from a Danish firm, Solural Pharma.
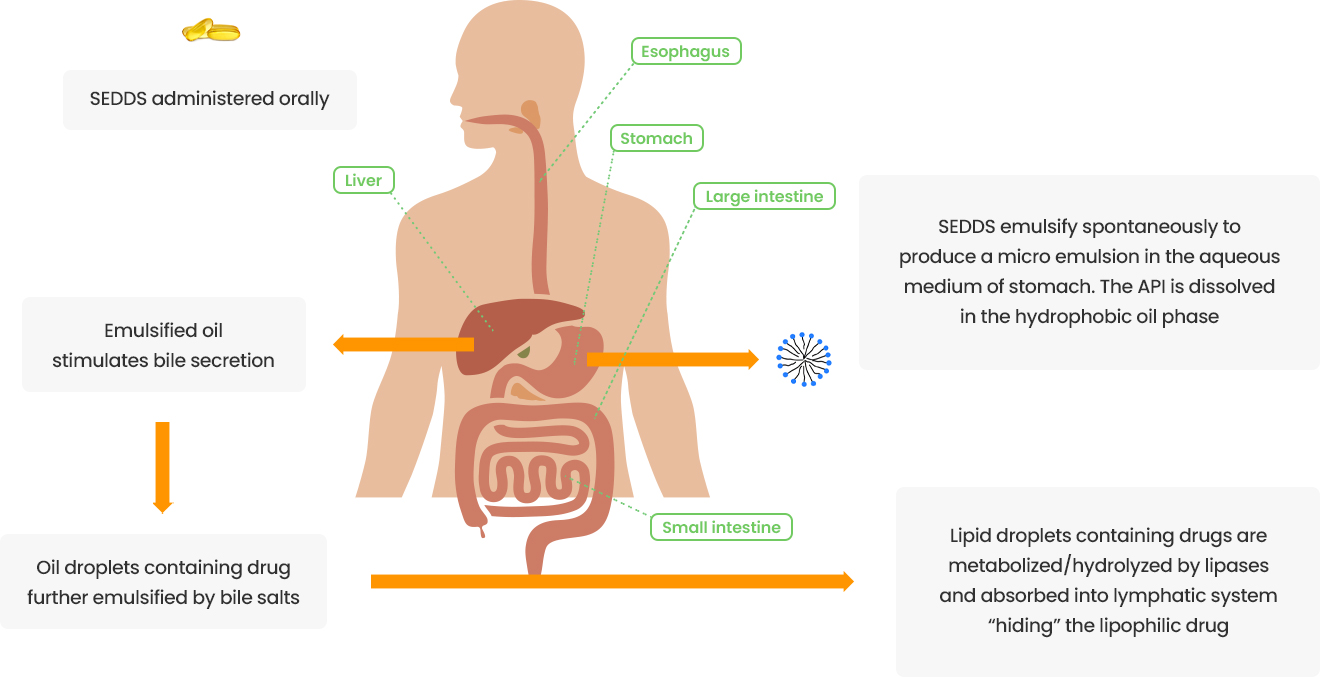
Existing prostate cancer treatments containing abiraterone acetate require administration at least 2 hours after a meal and prohibit food intake for at least 1 hour post-dose. To address this limitation, SOL-804 is being developed as an incrementally modified drug, utilizing the drug delivery platform ‘Lymphatic Targeting Technology™’. This approach not only mitigates the impact of food intake but also strives to reduce side effects through low-dose administration.
SOL-804 has successfully completed exploratory Phase 1 clinical trials, yielding significant pharmacokinetic findings when compared to existing treatments, particularly at low dosages. In December 2021, a composition patent was registered in the United States, further bolstering our intellectual property. Additionally, patent registrations have been completed in a total of 33 countries, including key nations such as Korea, Europe, Australia, Brazil, Canada, India, Japan, Mexico, and South Africa.